More Information
Submitted: October 13, 2022 | Approved: November 21, 2022 | Published: November 22, 2022
How to cite this article: Maffei G, Taurino L, Villani G, Rinaldi M, Cella A, et al. Contamination of a water bottle warmer in neonatal Intensive care unit and Klebsiella Pneumoniae ESBL + outbreak: cases series. Arch Pathol Clin Res. 2022; 6: 032-034.
DOI: 10.29328/journal.apcr.1001033
Copyright License: © 2022 Maffei G, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Klebsiella Pneumoniae ESBL +; Colonization; Newborn; Water warmer bottle
Contamination of a water bottle warmer in neonatal Intensive care unit and Klebsiella Pneumoniae ESBL + outbreak: cases series
Gianfranco Maffei1*, L Taurino1, G Villani1, M Rinaldi1, A Cella1, R Piemontese1, P Oliverio1, P Liberatore1, M Mancini1, G D’Angelo1, A Fracchiolla1, R Lamparelli1, R Di Tullio1, P Zaza1, R De Nittis2 and G Villone3
1Neonatology and Neonatal Intensive Care Unit, University Hospital, Foggia, Italy
2Clinical Pathology Department, University Hospital, Foggia, Italy
3Health Management, University Hospital, Foggia, Italy
*Address for Correspondence: Gianfranco Maffei, Neonatology and Neonatal Intensive Care Unit, University Hospital, Foggia, Italy, Email: [email protected]
Infections caused by extended-spectrum β-lactamases (ESBLs)-producing bacteria in particular Klebsiella Pneumoniae (KPE+), are on a constant rise and are a noted cause of outbreaks in neonatal intensive care units (NICUs). In the NICU of Policlinico di Foggia, an outbreak of infections in 2018 brought to the start of a serial check of presence and favorable conditions associated with KPE+ diffusion. The outbreak was controlled by improving basic hygiene measures such as hand washing, the use of disposable gloves and gowns, and removing a potential environmental contamination source such as the water bottle warmer.
Klebsiella pneumoniae is the Gram-negative most relevant opportunistic pathogen, causing nosocomial infections, especially among critical patients in the intensive care units [1]. Klebsiella pneumoniae naturally colonizes the nasopharyngeal and gastrointestinal tracts in 15% and 20% of patients respectively [2]. K. pneumoniae-producing extended-spectrum β-lactamases (ESBLs) (KPE+) are currently found throughout the world and have caused numerous outbreaks of infection [3]. Regular surveillance swabs for KPE+, hygiene measures and prevention of environmental contamination play a crucial role in contrasting the KPE+ spread.
In September 2018 there was an outbreak of K. pneumoniae. For this reason, we started submitting periodically all newborns admitted to NICU to nasopharyngeal and rectal swabs in order to investigate the presence of KPE+. Hygiene practices and the hospital environment were investigated too.
From September 2018 to September 2019 all newborns admitted to the Neonatology unit and NICU of Policlinico di Foggia (Italy), were submitted twice a week, to nasopharyngeal and rectal swabs, to assess the incidence of KPE+. The newborns with positive nasopharyngeal or rectal swabs were further studied through the dosage of C-reactive protein (CRP) in the blood and blood culture. The newborns who had at least a positive nasopharyngeal or rectal swab, without any clinical signs and with negative CRP and negative blood culture, were defined as “colonized”. Those who had at least a positive nasopharyngeal or rectal swab and the presence of clinical signs of infection associated with positive CRP and/or positive blood culture were defined as “infected”. Furthermore, in order to find a potential environmental contamination source, the water bottles warmer and the container for carrying baby bottles were screened for KPE+ too.
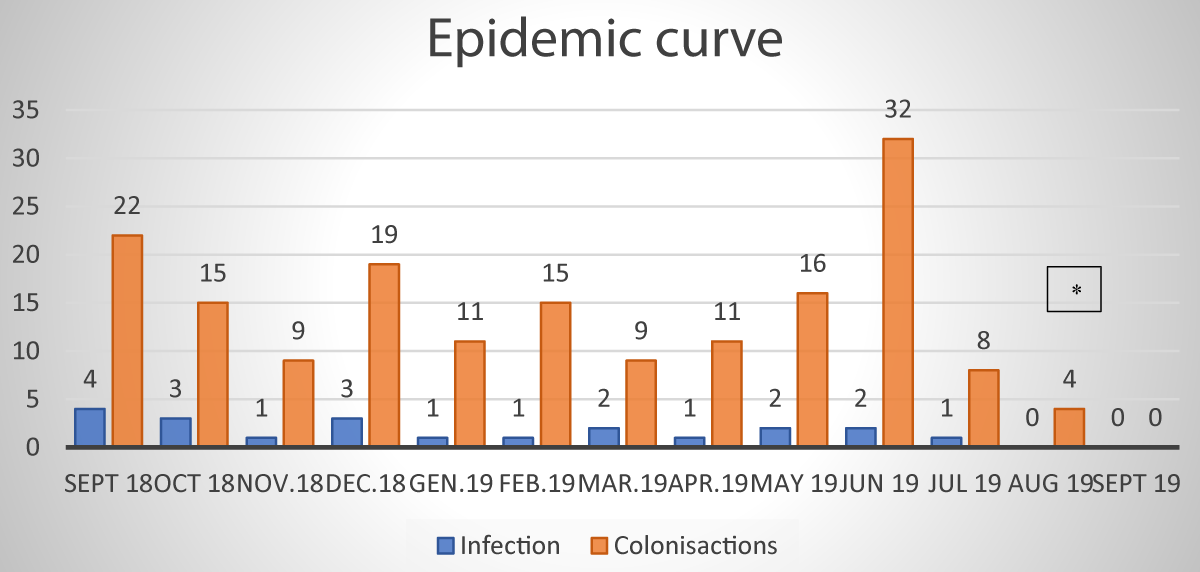
The patients admitted in Neonatology and NICU were 1028. The total number of surveillance swabs performed was 4122. One hundred and seventy-one patients resulted positive for one or both swabs. Among these, 25 were very low birthweight (VLBW) preterm, and 146 with weights greater than 1500 g. The median gestational age was: 29, 6 weeks (BW < 1500 g) and 36,6 weeks (BW > 1500 g). 150 newborns (15 VLBW and 135 with weight greater than 1500 g) were colonized and 21 newborns (2 VLBW and 17 with weight greater than 1500 g) were recorded. Of the 21 patients who presented septicemia, the infection developed from the respiratory tract, causing ventilatory status deterioration; the rest of them had elevated inflammatory indices and abdominal distension. 18 infected infants recovered after 10 days of treatment with two appropriate antibiotics given intravenously (meropenem and amikacin). 3 infants died from multiorgan failure No therapy was performed in the colonized patients who spontaneously came back negative. Finally, since the water bottle warmer resulted positive for KPE+ it was removed immediately (July 2019). Figure 1 shows the number of newly colonized and newly infected in the period under consideration.
Preventive measures Our protocol for infection prevention and control started in June 2019 and includes the following measures:
1) appropriate hand hygiene with different products including running water, liquid soap dispensers, and alcohol-based hand rubs placed in all rooms with a recommendation to healthcare givers to sanitize hands before and after coming in contact with patients; 2) disposable gloves for healthcare professionals when handling patients and if the situation (chest drainage placement, CVC placement, etc.) requires it, a protective face mask, cap, and disposable gowns; 3) all non-disposable equipment segregated and sterilized appropriately after every single usage.
In order to contrast the KPE+ outbreak, new preventive measures were implemented to limit transmission from patient to patient by healthcare workers.
Infected or colonized patients were grouped in the same area of the ward, isolated from the other patients, and cared for by specific nurses not involved with the care of negative patients.
Healthcare workers were trained in the prevention of nosocomial infections. It has been recommended they improve handwashing, use frequently hydro-alcoholic hand rubs, and to ware always disposable gloves and gowns during contact with patients.
Despite these measures, new cases occurred which prompted a search for an environmental cause of the outbreak.
Thus, swabs to the water taps in the ward and the caregivers’ hands (physician and nurse) were performed and resulted in a negative for the causative organism.
Finally, was proposed the hypothesis that the water bottle warmer was colonized and so it was immediately removed.
The effectiveness of this last measure was confirmed in October 2019 and January 2020 by biweekly screening with surveillance swabs to all NICU patients, which resulted in all negative for KPE+.
The 4 cases in August (Figure 1) were infants who had only rectal colonization which disappeared in the following days.
Figure 1: Number of newly colonized and newly infected in the period under consideration. (*) The 4 cases in August were infants who had only rectal colonization which disappeared in the following days.
Many Enterobacteriaceae outbreaks in NICU have been reported [4,5]. These organisms, present in the environment, become potentially invasive and highly virulent in very low birth weight preterm infants (< 28 postmenstrual weeks and birth weight < 1500 g). Patient-related factors (gestational age and birth weight) in our cases do not seem to play an important role in the prognosis of neonatal Klebsiella Pneumoniae infection. During a sudden large outbreak such as that reported here, the detection and prevention of patient-to-patient transmission are necessary but not sufficient. The detection of environmental contamination by painstaking detective work is also essential. The source of contamination must be looked for extensively, not only in the products administered to the newborns but also in their containers [6,7]. A large number of potential sources of contamination is a major difficulty [8,9]. Water bottle warmer contamination has not been never reported previously in the literature. Neonatologists should consider this possible source of contamination when several cases of life-threatening infection caused by the same Enterobacter occur in a NICU.
The infection committees of the hospitals can develop their protocols for the cleaning of water bottle warmer, or they can create them.
- Podschun R, Ullmann U. Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin Microbiol Rev. 1998 Oct;11(4):589-603. doi: 10.1128/CMR.11.4.589. PMID: 9767057; PMCID: PMC88898.
- Martin RM, Bachman MA. Colonization, Infection, and the Accessory Genome of Klebsiella pneumoniae. Front Cell Infect Microbiol. 2018 Jan 22;8:4. doi: 10.3389/fcimb.2018.00004. PMID: 29404282; PMCID: PMC5786545.
- Tumbarello M, Spanu T, Sanguinetti M, Citton R, Montuori E, Leone F, Fadda G, Cauda R. Bloodstream infections caused by extended-spectrum-beta-lactamase-producing Klebsiella pneumoniae: risk factors, molecular epidemiology, and clinical outcome. Antimicrob Agents Chemother. 2006 Feb;50(2):498-504. doi: 10.1128/AAC.50.2.498-504.2006. PMID: 16436702; PMCID: PMC1366869.
- Gras-Le Guen C, Lepelletier D, Debillon T, Gournay V, Espaze E, Roze JC. Contamination of a milk bank pasteuriser causing a Pseudomonas aeruginosa outbreak in a neonatal intensive care unit. Arch Dis Child Fetal Neonatal Ed. 2003 Sep;88(5):F434-5. doi: 10.1136/fn.88.5.f434. PMID: 12937053; PMCID: PMC1721613.
- Rettedal S, Löhr IH, Natås O, Giske CG, Sundsfjord A, Øymar K. First outbreak of extended-spectrum β-lactamase-producing Klebsiella pneumoniae in a Norwegian neonatal intensive care unit; associated with contaminated breast milk and resolved by strict cohorting. APMIS. 2012 Aug;120(8):612-21. doi: 10.1111/j.1600-0463.2012.02879.x. Epub 2012 Mar 24. PMID: 22779683.
- Donowitz LG, Marsik FJ, Fisher KA, Wenzel RP. Contaminated breast milk: A source of Klebsiella bacteremia in a newborn intensive care unit. Rev Infect Dis. 1981 Jul-Aug;3(4):716-20. doi: 10.1093/clinids/3.4.716. PMID: 7041217.
- Huerta-García GC, Miranda-Novales G, Díaz-Ramos R, Vázquez-Rosales G, Solórzano-Santos F. Intestinal Colonization by Extended-Spectrum Beta-Lactamase-Producing Enterobacteriaceae in Infants. Rev Invest Clin. 2015 Sep-Oct;67(5):313-7. PMID: 26696335.
- Reiss I, Borkhardt A, Füssle R, Sziegoleit A, Gortner L. Disinfectant contaminated with Klebsiella oxytoca as a source of sepsis in babies. Lancet. 2000 Jul 22;356(9226):310. doi: 10.1016/S0140-6736(00)02509-5. PMID: 11071189.
- Cadot L, Bruguière H, Jumas-Bilak E, Didelot MN, Masnou A, de Barry G, Cambonie G, Parer S, Romano-Bertrand S. Extended spectrum beta-lactamase-producing Klebsiella pneumoniae outbreak reveals incubators as pathogen reservoir in neonatal care center. Eur J Pediatr. 2019 Apr;178(4):505-513. doi: 10.1007/s00431-019-03323-w. Epub 2019 Jan 23. PMID: 30671695.